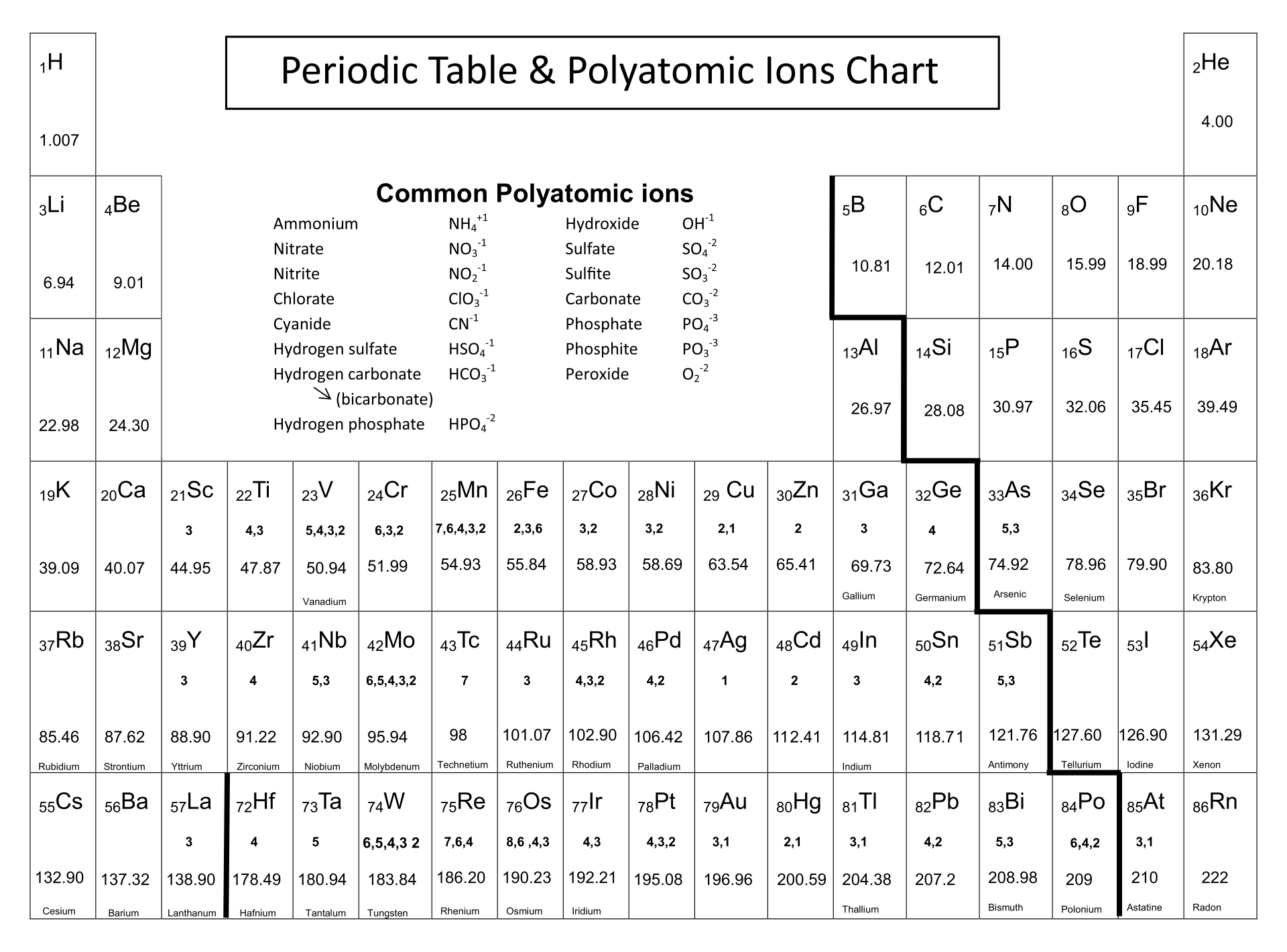
What characteristic charge do atoms in the third-to-last column of the periodic table have when they become ions?ġ3. The periodic table ionic charge can be broken down by metals that are positive and on the left of the table and nonmetals which are negative and found on. When a neutral atom gains or loses an electron, creating an anion or cation, the atoms radius. Neutral atoms tend to increase in size down a group and decrease across a period. One such trend is closely linked to atomic radii - ionic radii. What characteristic charge do atoms in the second column of the periodic table have when they become ions?ġ2. Common periodic trends include those in ionization energy, atomic radius, and electron affinity. What characteristic charge do atoms in the first column of the periodic table have when they become ions?ġ1. Using Lewis diagrams, show the electron transfer for the formation of CaF 2.ġ0. In many cases, the elements position on the periodic table will help you determine the kind of ion formed (anion or cation) and the size of the ionic charge. Using Lewis diagrams, show the electron transfer for the formation of Li 2O.ĩ. Science Notes Posts Contact Science Notes About Science Notes. Each entry contains the ions name, molecular formula and chemical structure. Using Lewis diagrams, show the electron transfer for the formation of MgO.Ĩ. This polyatomic ions list contains many common polyatomic ions grouped by charge. Using Lewis diagrams, show the electron transfer for the formation of LiF.ħ. Identify each as a cation, an anion, or neither.Ħ. Identify each as a cation, an anion, or neither.ĥ. When the following atoms become ions, what charges do they acquire?Ĥ. Use Lewis diagrams to illustrate the formation of an ionic compound from a potassium atom and an iodine atom.ģ.So, this makes the ion have a negative charge.\) By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. predict which element differs the most from. predict which element forms the most stable compounds in the +2 oxidation state. When we gain more electrons, we can know that the amount of electrons is higher than the proton. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. So, we can conclude that this ion has a positive charge and vice versa. So, the dominant charge of the certain ion is the proton. If the ion loses the electron, this means that the negative charge is lesser than the positive charge (proton). Actually, to understand the ion, we can see how the atoms lose their negative charge (electron). Actually, we have discussed this before above. When we download a periodic table of ions, we also need to know about how to determine an ion. Be sure to memorize silver (Ag+) and zinc (Zn2+) charges in addition to this chart. Within a group (family) of elements, atoms form ions of a certain charge. Thus, the periodic table becomes a tool for remembering the charges on many ions. Then, if it is reversed, which means the amount of electrons is higher than the proton, we can call it anion. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. The cation is where the positive charge (proton) has more amount than the electron. There are two kinds of ions in the periodic table of ions. However, it is pretty normal to be found that there is an imbalance of the charge, whether the number of protons is higher or the number of electrons is higher. Well, there are two kinds of ions on the periodic table.Īctually, if an atom or atoms that have equal numbers of negative charge (electrons) and positive charge (proton), then they can be defined as a neutral atom. Yes, one of the things that is required to be understood in the beginning is the ions themselves. The periodic table, its story and its significance. A neutral molecule that has an area of positive charge and an area of negative charge is called a zwitterion. An ion with a +2 charge is a cation that is also called a dication. IUPAC involvement covers various aspects. 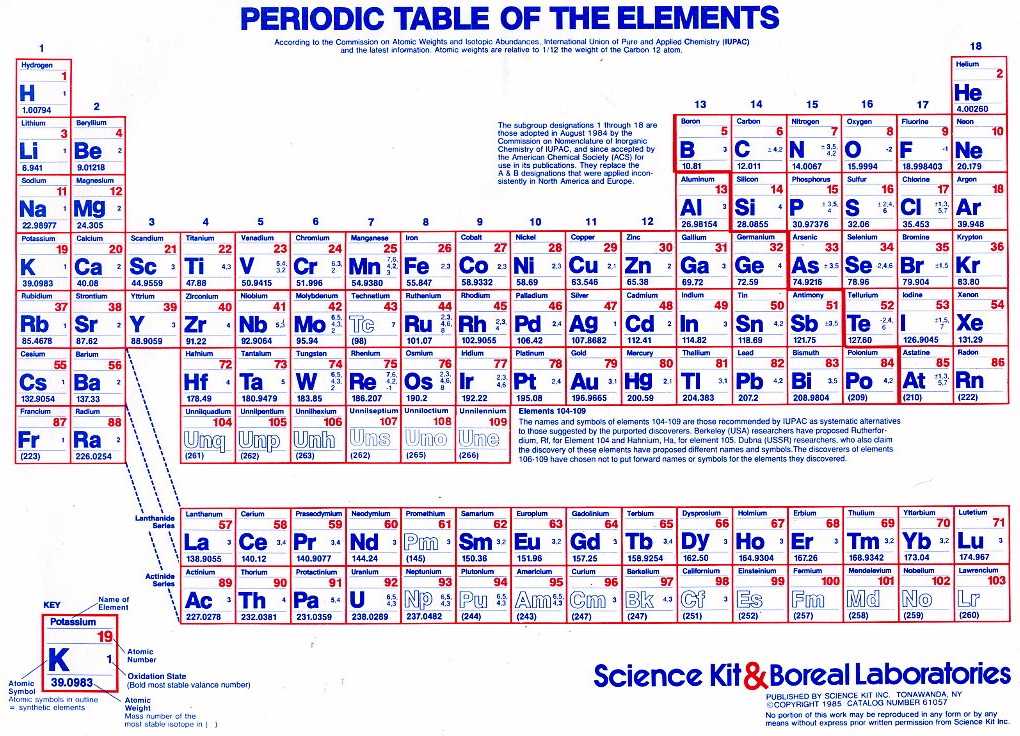
Yes, we can call this a periodic table of ions.īefore we download the periodic table of irons, we actually need to understand the basic things inside the periodic table of ions. An ion with a -2 charge is an anion that is also called a dianion. By virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. When it comes to learning Chemistry lessons, we may have seen our teacher introduce us with a sheet of the grid that consists of many abbreviations as a symbol of the elements. The nonmetals gain electrons until they have the same number of electrons as the nearest noble gas (Group 8A), forming negatively charged anions which have.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
